Home > Treatment Options > Tumor Suppressors > Clinical Trials >
Everolimus (Afinitor™)
Last Updated: 03/27/25
Status: Currently a NF2 Clinical Trial
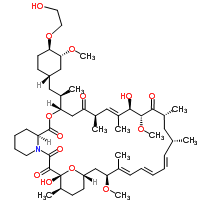
Trade Name:
Afinitor™ FermentekTM
Synonyms:
RAD-001, RAD001, Certican, Afinitor, Zortress, Sirolimus,
SDZ-RAD, RAD001, Everolimus [USAN], RAD 001, Rapamycin,
42-O-(2-hydroxyethyl)-, UNII-9HW64Q8G6G
SDZ-RAD, RAD001, Everolimus [USAN], RAD 001, Rapamycin,
42-O-(2-hydroxyethyl)-, UNII-9HW64Q8G6G
Pharmaceutical Company:
Novartis
Legal Status:
℞ - Trial
Administered By:
Pill - Oral
Tumor Target:
Vestibular Schwannoma
Protein Target:
mTOR Inhibitors - specifically mTORC1
Formula:
C53H83NO14
PubChem
ChemSpider
NF2 Trials
Recruiting: NCT01345136
NCT02352844
Active: NCT01490476 & NCT01880749
Completed: NCT01419639 &
NCT01024946
NCT02352844
Active: NCT01490476 & NCT01880749
Completed: NCT01419639 &
NCT01024946
Side Effects
Infections, anemia, anorexia, hyperglycemia, hyperlipidemia, headache, pneumonitis, epistaxis, GI disorders.
Most common reason of inability to remain on Everolimus was irritation of mucous membranes. Mucous membranes an epithelial tissue that secretes mucus and that lines many body cavities and tubular organs including the gut and respiratory passages. - mouth or on the genitals.
Common Side Effects
- Lung and Breathing Problems
- Infections
- Kidney Failure
- Mouth and Vaginal Ulcers
- Liver problems
- Vaccinations - patients should not have live vaccinations while on Afinitor
- Contraindicated with certain medications and certain foods
Additional Possible Side Effects:
- Feeling Weak
- Cough
- Shortness of Breath
- Diarrhea
- Rash, Dry Skin, Itching
- Nausea and Vomiting
- Fever
- Loss of Appetite
- Swelling - Arms, Legs, Feet or Ankles
- Infections - Respiratory Tract, Sinuses and Ears
- Constipation
- Stomach pain
- Seizure
- Headache
- Dizziness
- Stuffy / Runny Nose
- change in personality
- Low White Cell Count
- High Fat Content in Blood
Other Uses
- Advanced Kidney Cancer
- Renal Transplantorgan Rejection Prevention
- Subependymal Giant Cell Astrocytoma (SEGA)
- Progressive / Metastatic Pancreatic Neuroendocrine Tumors
- Breast Cancer
Notes:
Everolimus or similar may be neded for management of skin irritation
Sources
- https://www.cancerresearchuk.org/about-cancer/treatment/drugs/everolimus
- Salido-Vallejo, R., G. Garnacho-Saucedo, and A. Velez. "Elucidation of the mTOR pathway and therapeutic applications in dermatology." Actas Dermo-Sifiliograficas (English Edition) 107.5 (2016): 379-390. Source: http://www.sciencedirect.com/science/article/pii/S1578219016300154 | DOI: https://doi.org/10.1016/j.adengl.2016.03.001
- Karajannis, Matthias A., et al. "Phase II study of everolimus in children and adults with neurofibromatosis type 2 and progressive vestibular schwannomas." Neuro-oncology 16.2 (2014): 292-297. Source: https://academic.oup.com/neuro-oncology/article/16/2/292/1084065 | DOI: https://doi.org/10.1093/neuonc/not150


 |Google Play
|Google Play