Home > Treatment Options > Tumor Suppressors > Clinical Trials >
Octreotide (Sandostatin)
Last Updated: 01/31/18
Status: NF2 Trial in only Turkey at this time
Index
- Basic Information
- Side Effects
- Other Uses
- Additinal Information
1. Basic Facts
| Other Names | Octreotida, Octreotidum, Octrotide, SMS 201-995 PA, SMS995-AAA, SMS 201-995 AC, Octreotide Hydrochloride, Sandostatin, Octreotide Pamoate, Sandostatin, Octreotide Acetate, SMS-201-995 AC, Sandostatin Lar, Octreotide, SMS-201-995, SMS PA |
|---|---|
| Trade Name | Sandostatin™ FermentekTM, , Sandostatin, and Sandostatin Lar |
| Tumor Target | Vestibular Schwannoma |
| Treatment By | IV or Port |
| Inhibitor | |
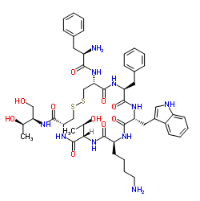
| Molecular Formula | C49H66N10O10S2 |
| IUPAC Name | |
| PubChem | |
| ChemSpider |
2. Side Effects:
"The most frequent adverse effects (more than 10% of patients) are headache, hypothyroidism, cardiac conduction changes, gastrointestinal reactions (including cramps, nausea/vomiting and diarrhea or constipation), gallstones, reduction of insulin release, hyperglycemia or hypoglycemia, and (usually transient) injection site reactions. Slow heart rate, skin reactions such as pruritus, hyperbilirubinemia, hypothyroidism, dizziness and dyspnea are also fairly common (more than 1%). Rare side effects include acute anaphylactic reactions, pancreatitis and hepatitis. One study reported a possible association with rheumatoid arthritis."
"Some studies reported alopecia in patients who were treated by octreotide. Rats which were treated by octreotide experienced erectile dysfunction in a 1998 study."
"A prolonged QT interval has been observed in patients, but it is uncertain whether this is a reaction to the drug or part of the patients' illnesses."
3. Other Uses
Currently in trials for; Breast Carcinoma, Diarrhea, Carcinoid Tumor, Carcinoid Tumor, Hepatocellular Carcinoma, Carcinoid Syndrome, Diabetic Retinopathy, and Pancreatic Carcinoma.
https://clinicaltrials.gov/ct2/show/NCT00001860 Last Update Posted: March 4, 2008https://clinicaltrials.gov/ct2/show/NCT00050635 November 19, 2009
4. Additinal Information
Octreotide is the acetate salt of a cyclic octapeptide. It is a long-acting octapeptide with pharmacologic properties mimicking those of the natural hormone somatostatin.


 |Google Play
|Google Play