Home > Treatment Options > Tumor Suppressors > Clinical Trials >
Lapatinib (Tykerb™)
Last Updated: 01/29/20
Status: Currently a NF2 Clinical Trial
Trade Name:
Tykerb™
Synonyms:
Tykerb, Lapatinib Ditosylate, GW572016, 231277-92-2,
GW 572016, Tycerb, 1xkk, Lapatinib (INN), Lapatinib [INN]
GW 572016, Tycerb, 1xkk, Lapatinib (INN), Lapatinib [INN]
Pharmaceutical Company:
GlaxoSmithKline
Legal Status:
℞ - Prescription Only
Administered By:
Pill - Oral
Tumor Target:
Vestibular Schwannoma
Protein Target:
TKI and EGFR Inhibitors
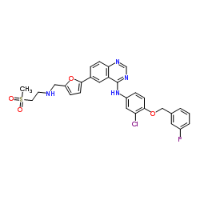
Formula
C29H26ClFN4O4S
ChemSpider
PubChem
NF2 Trials:
Recommendation
Lapatinib causes fatigue for several hours, medication should be taken at nighttime before sleep, not in the morning.
Other Known Uses
Breast cancer and tumors developed Solid Tumor.
Side Effects
Minimal Side Effects
Major side effects are uncommon. Since people with NF2 are taking these medications at a low dose, only minimal side effects are common. However, long term use is primarily where these major side effects take place. Monthly Blood and Urine Tests can usually point to an issue before it's a real problem. Talk to your doctor.
- Memory Loss
- Acne
- Nail Infections
- Vision - Light Sensitivity
Additional Side Effects to Watch for:
- Liver Damage
- Anemia
- Low White Cell Count
- Low Platlet Count
- Risk of Infection
- Nausea
- Vomiting
- Diarrhea
- Belly Pain
- Skin Irritation
- Rash
- Mouth Irritation
- Numbness / Tingling of Hands and Feet
- Fatigue
- Irregular Menstrual Cycle
4. Warning:
Liver Damage: Look for dark urine, feeling tired, not hungry, nausea, vomiting or yellowing of skin and/or eyes.


 |Google Play
|Google Play