Home > Treatment Options > Tumor Suppressors > Clinical Trials >
Crizotinib (Xalkori®)
Status: Available in 2019 for NF2 in the United States and Canada
Trade Name:
Xalkori™
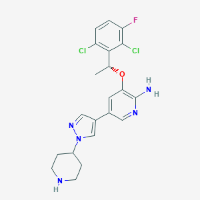
Chemical Compound:
Crizotinib
Synonyms:
CRZ, PF-2341066, PF-02341066,
PF2341066, PF-2341066, PF 2341066
PF2341066, PF-2341066, PF 2341066
Pharmaceutical Company:
Pfizer
Legal Status:
℞ - Prescription
Administered By:
Pill - Oral
Tumor Target:
Schwannoma
Protein Target:
FAK1 (PTK2) Inhibitor
NF2 Trials:
Recruiting Trial: NCT02465060
Formula:
C21H22Cl2FN5O
Helpful Facts
- In trials for: Breast Cancer, solid tumor cancer
- Solubility: Soluble in DMSO, not in water. Dimethyl Sulfoxide (DMSO) is an organosulfur compound with the formula (CH3)2SO.
- Mammalian target of rapamycin (mTOR) serine/threonine kinase (dual mTOR Complex 1 and 2 inhibitor, mTORC1 and mTORC2)
- Inhibitor: inhibits the membrane receptor MET and activation of the MET signaling pathway
Side Effects
- upper respiratory infection,
- nausea,
- vomiting
- stomach pain,
- decreased appetite,
- insomnia,
- dizziness,
- tired feeling,
- diarrhea,
- constipation,
- rash or itching,
- cold symptoms (stuffy nos
- swelling in hands or feet.
Tell your doctor if you have serious side effects of Xalkori including:
- severe dizziness, fainting, fast or pounding heartbeats;
- vision problems such as blurred vision, increased sensitivity of your eyes to light, or seeing flashes of light or "floaters";
- chest pain, dry cough or cough with mucus, wheezing, shortness of breath;
- easy bruising, unusual bleeding, purple or red pinpoint spots under your skin;
- fever, chills, body aches, flu symptoms, sores in your mouth and throat;
- loss of appetite, dark urine, clay-colored stools, or jaundice (yellowing of the skin or eyes).
Sources
- Rx Lise. "Xalkori." Last reviewed on RxList 7/8/2019 https://www.rxlist.com/xalkori-side-effects-drug-center.htm
-
Zhao, Yingchao, et al. "Targeting the cMET pathway augments radiation response without adverse effect on hearing in NF2 schwannoma models."
Proceedings of the National Academy of Sciences (2018): 201719966.
Source: http://www.pnas.org/content/early/2018/02/07/1719966115 | DOI: 10.1073/pnas.1719966115 -
Troutman, Scott, et al. "Crizotinib inhibits NF2-associated schwannoma through inhibition of focal adhesion kinase 1."
Oncotarget 7.34 (2016): 54515.
Source: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5342359/ | DOI: 10.18632/oncotarget.10248 - National Center for Biotechnology Information. PubChem Compound Database; "CID=11626560", (accessed Jan. 12, 2018) Source: https://pubchem.ncbi.nlm.nih.gov/compound/11626560
-
MedKoo Biosciences. "Crizotinib - MedKoo CAT#: 202222" (accessed Jan. 12, 2018)
Source: http://www.medkoo.com/products/4537
-
U.S. Food and Drug Administration. "FDA expands use of Xalkori to treat rare form of advanced non-small cell lung cancer." (March 11, 2016)
Source: https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm490329.htm - Troutman, Scott, et al. "Crizotinib inhibits NF2-associated schwannoma through inhibition of focal adhesion kinase 1." Oncotarget 7.34 (2016): 54515. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5342359/


 |Google Play
|Google Play