Home > Treatment Options > Tumor Suppressors >
BXCL101 (Bortezomib™)
Last Updated: 02/16/18
Status: As of 01/18, NF2 Clinical Trial Have Not Started
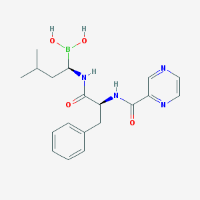
| Trade Names: | Bortezomib™, Velcade™, and Neomib™ |
|---|---|
| Chemical Compound: | BXCL101, Bortezomib, PS-341 |
| Synonyms: | PS-341, LDP 341, LDP-341, LDP341, MLN341, ps 341, PS 341, 341 |
| Pharmaceutical Companies: | BioXcel Therapeutics, previously Millennium Pharmaceuticals |
| NF2 Trial Availability | Initially Proposed to start in the US before 2018 |
| Legal Status: | ℞ - Prescription Only |
| Administered By: | Intravenous (IV) or Port |
| Tumor Target: | Schwannoma and Meningioma | Protein Target: | Proteasome Inhibitor |
| Formula: | C19H25BN4O4 |
| NF2 Trials | Active, Not Recruiting: University of Virginia |
Chemical Compound BXCL101 was designed by Myogenics Pharmecuticles initially under treatment called Velcade™ in the United Kingdom (UK). It proved to have help Multiple Myeloma and Mantle Cell Lymphoma. In 2006, Velcade™ was rejected by the UK's 'National Institute for Health and Care Excellence' (NICE), an executive non-departmental public body of the Department of Health in the United Kingdom due to cost, not an issue of effectiveness.
Other Conditions
Bortezomib has been in trial in the United States for Multiple Myeloma and Mantle Cell Lymphoma. Like the UK trials, the trials listed under ClinicalTrials.org, are Combination trials which include Dexamethasone
Possible Side Effects
Common Side Effects
- mild nausea, vomiting, upset stomach;
- diarrhea, constipation, bloating;
- headache, mild dizziness;
- muscle pain, bone or joint pain;
- sleep problems (insomnia);
- mild rash or itching;
- skin irritation where the medicine was injected
Serious Side Effects
- easy bruising or bleeding;
- tingling/numbness/pain/burning feeling of the hands or feet;
- fainting;
- stomach pain;
- black stools;
- coffee-ground vomit;
- trouble breathing;
- swelling or pain in the lower legs;
- fast or irregular heartbeat;
- severe headache;
- vision problems;
- mental/mood changes (e.g., rarely, thoughts of suicide);
- swelling of the hands/ankles/feet;
- changes in the amount of urine;
- yellowing skin or eyes;
- dark urine
Trials
- "Bortezomib and Temozolomide in Treating Patients With Brain Tumors or Other Solid Tumors That Have Not Responded to Treatment"
https://clinicaltrials.gov/ct2/show/NCT00544284
Recruitment Status : Completed
First Posted : October 16, 2007
Last Update Posted : February 15, 2013
Sponsor: City of Hope Medical Center | Collaborator: National Cancer Institute (NCI)
Information from PubChem
"Bortezomib mainly causes sensory peripheral neuropathy, but severe motor peripheral neuropathy also has been reported. In the phase III trial, peripheral neuropathy occurred in thirty-six percent (36%) of patients receiving bortezomib and nine percent (9%) of patients receiving dexamethasone. About 8% of patients discontinued bortezomib therapy because of peripheral neuropathy."
"Patients receiving bortezomib should be monitored for manifestations of neuropathy (eg, burning sensation, hyperesthesia, hypoesthesia, paresthesia, discomfort, neuropathic pain). Dose and/or frequency of administration of bortezomib should be adjusted in patients who experience new-onset or exacerbation of peripheral neuropathy."
"Hypoglycemia and hyperglycemia have been reported in patients with diabetes mellitus who received bortezomib concomitantly with oral antidiabetic agents. If bortezomib is used concomitantly with oral antidiabetic agents, blood glucose concentrations should be monitored carefully and dosage of the antidiabetic agent adjusted as necessary."
"Potential interaction (increased risk of hypotension) when bortezomib is used with drugs that can cause hypotension. Dosage adjustment of hypotensive agents may be necessary."
Sources
- Ganjoo, Anita. "BioXcel's BXCL101, Receives Orphan Drug Designation from the U.S. FDA for the Treatment of Patients with Neurofibromatosis Type 2 (NF2)." GlobeNewswire News Room (September 2016) http://globenewswire.com/news-release/2016/09/13/871507/0/en/BioXcel-s-BXCL101-Receives-Orphan-Drug-Designation-from-the-U-S-FDA-for-the-Treatment-of-Patients-with-Neurofibromatosis-Type-2-NF2.html
- "Medical Definition of Antineoplastic." MedicineNet, WebMD, (May 2016) http://www.medicinenet.com/script/main/art.asp?articlekey=22631
- "Bortezomib" National Center for Biotechnology Information. PubChem Compound Database; CID=387447, https://pubchem.ncbi.nlm.nih.gov/compound/387447
- Cunha, John P. "Common Side Effects of Velcade (Bortezomib) Drug Center." RxList (December 2016) http://www.rxlist.com/velcade-side-effects-drug-center.htm
- ClinicalTrials.gov. www.ClinicalTrials.gov
Sources from 2018
-
Mateos, María-Victoria, et al. "Daratumumab plus Bortezomib, Melphalan, and Prednisone for Untreated Myeloma."
New England Journal of Medicine. (2017). http://www.nejm.org/doi/full/10.1056/NEJMoa1714678
"Among patients with newly diagnosed multiple myeloma who were ineligible for stem-cell transplantation, daratumumab combined with bortezomib, melphalan, and prednisone resulted in a lower risk of disease progression or death than the same regimen without daratumumab.


 |Google Play
|Google Play